Abstrakt
Açıq arterial axıntı kimi anadangəlmə ürək xəstəliyi olan xəstələrdə koronar arteriyalarda diastolik qan axınının pozulması səbəbindən yavaş koronar axının inkişaf riski artır. Yavaş koronar axın, obstruktiv lezyonlar olmadığı təqdirdə kontrastın koronar arteriyalardan gec keçməsi ilə xarakterizə olunan angioqrafik bir tapıntıdır. Əhəmiyyətli stenozun olmamasına baxmayaraq, yavaş koronar axın miokard işemiyasına səbəb ola bilər və mədəcik aritmiyaları da daxil olmaqla ciddi ağırlaşmalar riskini artıra bilər. İlk dəfə 1972-ci ildə təsvir edilən bu fenomen hələ də kifayət qədər öyrənilməmişdir. Bu klinik hal, hərtərəfli diaqnostik və terapevtik yanaşma tələb edən mədəcik aritmiyasının inkişafı ilə mürəkkəbləşən açıq arterial axıntı ilə əlaqəli yavaş koronar axını olan bir xəstəni təqdim edir.
Əsas mətn
Case report:
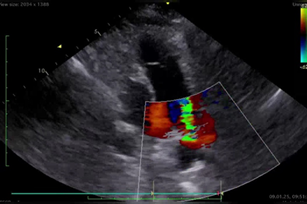
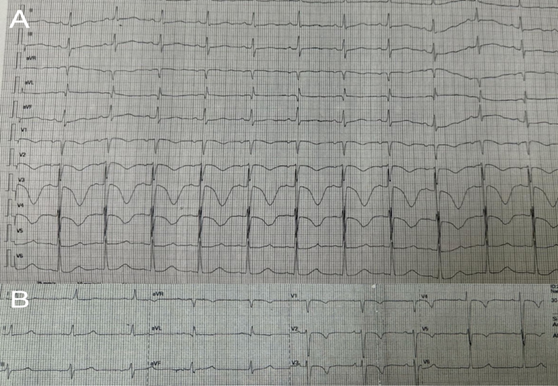
A 55-year-old woman was admitted to our clinic with complaints of palpitations, fatigue, and dyspnea (NYHA class 2). An ECG revealed sinus rhythm and biphasic T waves in leads I, AVL, and V6, QTc 390 msec, heart rate (HR) of 70 beats per minute (Fig. 1). The anamnesis included paroxysmal episodes of atrial fibrillation for many years. Transthoracic echocardiography (TTE) showed a normal left ventricular ejection fraction (LVEF 55%), a patent ductus arteriosus (PDA) with a duct diameter of 6 mm, and enlargement of the left ventricle and atrium (LVDD 62mm, LVSD 47mm, LAVI 44 ml/m2). There was moderate mitral regurgitation, mild tricuspid regurgitation, and dilation of the pulmonary artery. The pulmonary artery systolic pressure was 50 mmHg (Fig. 2). It was recommended that the PDA be closed using a percutaneous method. Both cardiac catheterization and coronary angiography were performed prior to the procedure. Cardiac catheterization revealed pulmonary artery pressure of 64/35/50 mmHg. A prominent patent ductus arteriosus was observed between the left pulmonary artery and the aorta. Coronary angiography did not show any occlusions in the coronary arteries, although extremely slow dye flow was observed (TIMI I). Based on these findings, the patient was prescribed a β-blocker, an SGLT-2 inhibitor, and continued anticoagulant therapy. Two weeks later, she was readmitted in our clinic with worsening dyspnea (NYHA class III-IV) and pronounced palpitations. An ECG revealed atrial fibrillation with high ventricular response. TTE showed a depressed left ventricular ejection fraction with global hypokinesia (LVEF 40-45%). On physical examination, blood pressure was 110/70 mmHg, and the HR was 140 - 150 beats per minute. Heart sounds were faint, with a grade 3/6 machine-like murmur at the upper left sternal border, and moist rales in the lower lung fields. Initial work up. The patient was hospitalized with a diagnosis of atrial fibrillation and heart failure. She had been taking rivaroxaban for more than one month, and amiodarone infusion (900 mg) was started for rhythm control. The day after hospitalization, sudden cardiac arrest occurred, and ventricular fibrillation was recorded on the monitor. CPR was immediately initiated according to recommendations, and cardioversion (220 J) was performed. Within a short time (3 minutes), the patient's vital signs were restored, and sinus rhythm was reestablished. The ECG showed sinus rhythm with significant prolongation of the QT interval (600 msec, QTc 629 msec), HR of 66 beats per minute, and a negative T wave in leads V1-V4 (Fig 3A). TTE revealed a further reduction in ejection fraction (LVEF 30%) after CPR. In the intensive care unit, the amiodarone infusion was discontinued, and intravenous therapy with magnesium sulfate and metoprolol was initiated. Trimetazidine was also prescribed to improve coronary microcirculation and myocardial metabolism. Complete blood count, troponin I level, serum electrolytes, creatinine, albumin, TSH, and INR were within normal limits. Elevated levels of liver enzymes (ALT 2102 U/L, AST 1734 U/L) and NT-proBNP (19,899 pg/mL) were noted.
Diagnosis and management
After the patient’s condition stabilized, cardiac MRI was performed to assess myocardial disease that could cause heart failure and arrhythmias. Enlargement of the left ventricle with reduced systolic function was noted (LVEDV 114 mL/m², LVESV 76 mL/m², LVEF 34%), along with moderate left atrial dilatation (33 cm²). Right heart chambers and right ventricular systolic function were within normal limits. T1 mapping showed prolonged T1 relaxation time and increased extracellular volume (T1 1053 ms, ECV 33%), with no evidence of late gadolinium enhancement or other ischemic/infiltrative changes. These findings were consistent with the effects of tachysystolic arrhythmia and the presence of a PDA.The follow-up ECG showed sinus rhythm with a normal QT interval (QTc 440 msec), a HR of 60 beats per minute, and a negative T wave in leads V1-V5 (Fig. 3B). Subsequently, the PDA was closed using a percutaneous approach. Medical treatment was initiated, including Valsartan/Sacubitril 24/26 mg, Spironolactone 25mg, Metoprolol 25mg, Rivaroxaban 20mg, Trimetazidine 80mg, Empagliflozin 10 mg, and Finerenone 10 mg.
At the follow-up examination after two months, the ECG showed sinus rhythm with no significant changes. The patient had no complaints and was classified as NYHA Class I. TTE revealed an improvement in the LVEF 50%, a reduction in the size of the left ventricle and atrium (LVDD 52 mm, LVSD 35 mm, LAVI 32 ml/m2), and mild mitral regurgitation and mild tricuspid regurgitation.
Discussion:
The present case describes a rare and clinically significant combination of patent ductus arteriosus and slow coronary flow complicated by malignant ventricular arrhythmia. Although PDA is a well-known cause of heart failure and atrial arrhythmias in adults, its association with SCF and subsequent ventricular fibrillation has been insufficiently explored. In this patient, coronary angiography revealed markedly delayed contrast progression (TIMI I flow) in the absence of obstructive coronary artery disease. This finding is consistent with SCF, which has been attributed to coronary microvascular dysfunction, increased small-vessel resistance, and impaired diastolic coronary perfusion. In the context of PDA, continuous left-to-right shunting and reduced aortic diastolic pressure may further compromise coronary blood flow, predisposing the myocardium to ischemia despite angiographically normal epicardial arteries. Several studies have demonstrated that SCF is associated with myocardial ischemia, ventricular repolarization abnormalities, QT interval prolongation, and an increased incidence of ventricular arrhythmias. In our patient, the development of ventricular fibrillation was preceded by QT prolongation, which was likely multifactorial. Potential contributing mechanisms include myocardial ischemia related to SCF, tachycardia-induced cardiomyopathy, electrolyte-independent repolarization disturbances, and the proarrhythmic effect of amiodarone in a patient with underlying QT vulnerability. The normalization of the QT interval after discontinuation of amiodarone and stabilization of hemodynamics further supports this hypothesis.
Cardiac MRI findings excluded ischemic scar or infiltrative cardiomyopathy and demonstrated diffuse myocardial changes compatible with tachyarrhythmia-related myocardial dysfunction and chronic volume overload due to PDA. Importantly, closure of the PDA combined with optimized heart failure therapy resulted in significant reverse remodeling, recovery of left ventricular systolic function, and complete clinical improvement, emphasizing the causal role of hemodynamic overload and impaired coronary microcirculation in this case. This case underscores several important clinical considerations. First, SCF should be recognized as a potentially serious condition rather than a benign angiographic finding, particularly in patients with congenital heart disease. Second, careful selection of antiarrhythmic therapy is crucial, as drugs that prolong the QT interval may increase the risk of malignant ventricular arrhythmias in patients with SCF. Finally, comprehensive management addressing both the underlying structural defect and coronary microcirculatory dysfunction is essential for optimal outcomes.
Conclusions
The goal of this case is to highlight the importance of not only correcting a patent ductus arteriosus but also considering the possibility of slow coronary flow. Slow coronary flow can contribute to the development of ventricular arrhythmias, especially in patients with congenital heart defects. It may indicate problems with coronary microcirculation and repolarization, which can lead to QT interval prolongation and dangerous arrhythmias. When choosing antiarrhythmic drugs, it is important to avoid those that can worsen QT prolongation, such as amiodarone. Instead, medications like trimetazidine that support heart metabolism and microcirculation may be preferred. Patients with slow coronary flow need regular ECG monitoring and careful treatment planning.
Şəkillər
Açar sözlər
İstinadlar
Məqalə barədə təfərrüatlar:
Nəşr tarixçəsi
Dərc edilib: 28.Jun.2025
Müəllif hüququ
© 2013-2025. Azərbaycan Kardiologiya Cəmiyyətinin rəsmi nəşri. Jurnal "Uptodate in Medicine" tibb nəşriyyatı tərəfindən dərc olunur. Bütün hüquqlar qorunur.Əlaqəli məqalələr
Baxılıb: 11


